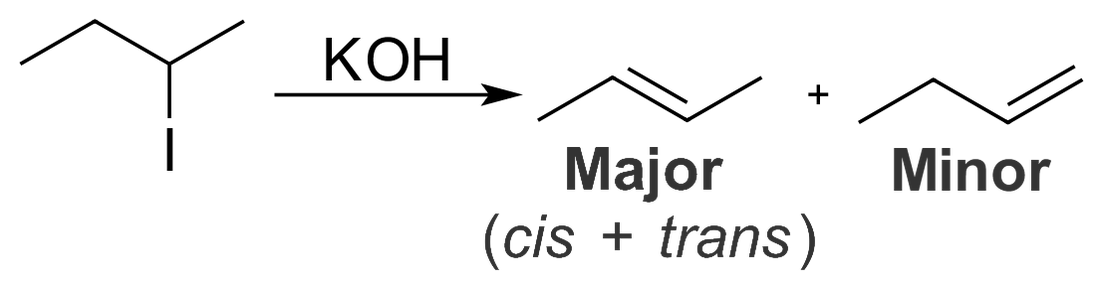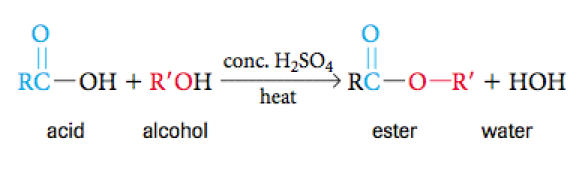Types of Organic Reactions
Addition Reactions
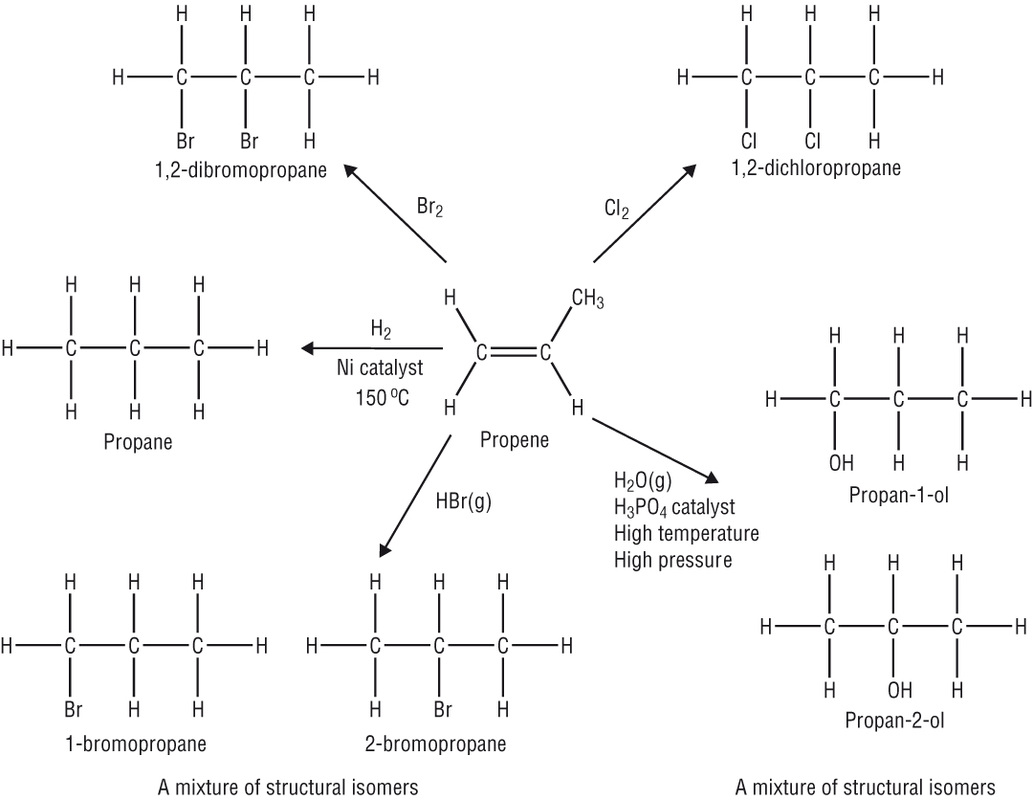
Addition reactions are which atoms or molecules are added to double or triple bonded carbon molecules to break up the bond to smaller bonds instead of having the double or triple carbon bond. These atoms are added to alkenes or alkynes, double and triple bonds respectively. It is a very similar concept to the idea of saturating or hydrogenating fat molecules.
Starting from the original form of propene it is possible to see the different forms that an addition reaction can take place.
- When 2 hydrogens are reacted, it forms an alkane
- When a halogen reacts with it, it forms an alkylhalide.
- When water reacts with it it forms an alcohol.
- When to halogens (halogen gas) reacts with it, it forms a dihaloalkane.
If a limited amount of a reagent (not the alkyne) is present then the alkyne will turn into an alkene. However, if there is excess reagent then it will form an alkane. Another example of deviation is when there is space for more than one molecule. In this case, then the molecule may have isomers after the reaction happens.
An example of this being used practically is when the addition of bromine is used with an alkene or alkyne to test the presence of double of triple bonds. At this point if there is a double of triple bond, the bromine will react and will lose its brownish color. However, if there are no bonds then the color will stay the same.
An example of this being used practically is when the addition of bromine is used with an alkene or alkyne to test the presence of double of triple bonds. At this point if there is a double of triple bond, the bromine will react and will lose its brownish color. However, if there are no bonds then the color will stay the same.
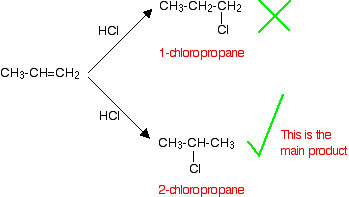
Markovnikov's Rule
In the addition of an H-X to an alkene, then the hydrogen will attach itself to the carbon with more hydrogen and the X will bind itself to the carbon with less hydrogen's attached.
Elimination Reactions
Elimination reactions are the reciprocal of the addition reaction in which instead of breaking apart alkene and alkyne bonds, they are formed instead. It is from 1 reactant to become 2 reactants instead. As an example alcohols undergo elimination reactions in the presence of strong acids such as sulfuric acid. It is the same as making a fat unsaturated from a state of saturation.
Zaitsev's Rule
In cases where a hydrogen can be removed from either side of the alkylhalide bond, the hydrogen will be removed most likely from the carbon that has the most carbon-carbon bonds and will form the major product. The hydrogen on the side with the lesser carbon-carbon bonds will from the minor product. This is known as Zaitsev's Rule.
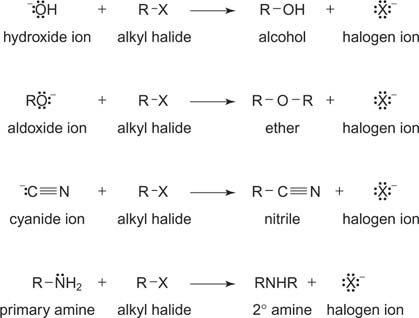
Substitution Reactions
This reaction is simply the switching of atoms on the carbon chain, thus its name of substitution. Two features can be found in this:
The general rule to the substitution reaction is that the A-Z will turn into A-Y after the reaction because the Z has switched with the Y.
- Two compounds react to form two different compounds
- carbon atoms are bonded to the same number of atoms in the product as in the reactant.
The general rule to the substitution reaction is that the A-Z will turn into A-Y after the reaction because the Z has switched with the Y.
Special cases are that when alcohols and haloalkanes under substitution reactions, alcohols as reactants will turn into a alkylhalide and produce water. With the alkylhalide as the reactant along with a hydroxide ion, it will form an alcohol and an ion.
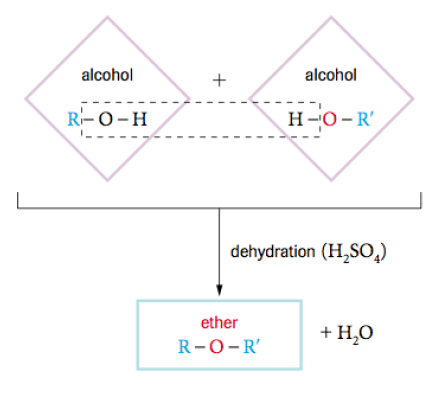
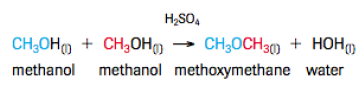
Condensation Reactions
This reaction is which two larger molecules combine to form one larger molecule and precipitate water out. It is common in the human body and it's systems and is commonly found between a carboxylic acid an an amine. It is also commonly seen in alcohols. By definition it is a reaction in which one molecule donates a hydroxide ion and the other molecule donates a hydrogen ion in order to form water an one large molecule.
Esterfication Reactions
Esterfication is under the umbrella of a condensation reaction in which is a name specially given to those reactions that involve a carboxylic acid an an alcohol. This reaction will yield an ester and water. Below is the formula for such a reaction.
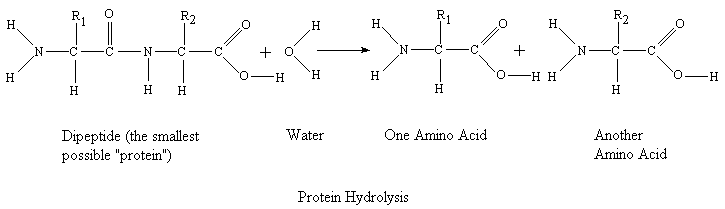
Hydrolysis Reactions
This is another type of reaction that is under the umbrella of a condensation reaction. It is actually the reverse of a condensation reaction in which water is added as a reactant to form 2 molecules from one molecule. It will usually form a sodium salt of the carboxylic acid. The hydroxyl group is added to the side of a bond with the ester or amide group and the hydrogen is added to the other side of the molecule.
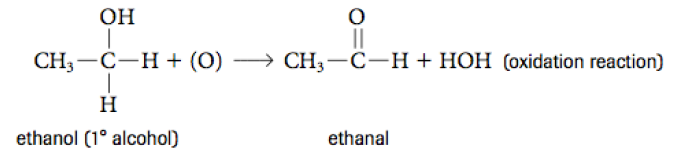
Oxidation Reactions
An oxidation reaction is one where there is a loss of electrons. It is one where a carbon atom forms more bonds to the oxygen.
The most important thing to remember is that an alcohol, when oxidized, turns into an aldehyde or ketone and an aldehyde when oxidized further turns into a carboxylic acid.
An oxidation is shown when the reactant side shows an oxygen within brackets.
The most important thing to remember is that an alcohol, when oxidized, turns into an aldehyde or ketone and an aldehyde when oxidized further turns into a carboxylic acid.
An oxidation is shown when the reactant side shows an oxygen within brackets.
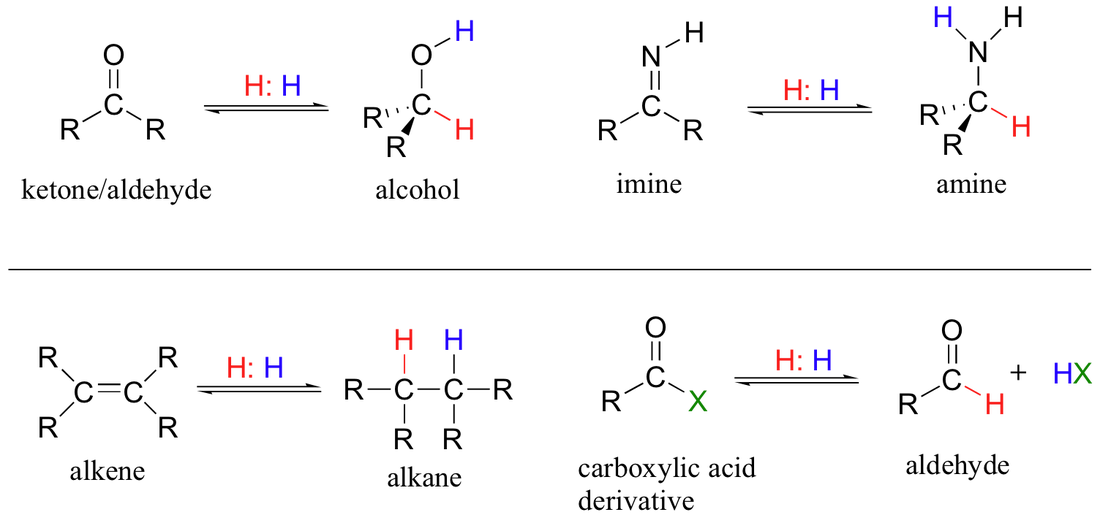
Reduction Reactions
A reduction reaction is one where there is a gain in electrons. It is one where a reaction in which a carbon atom forms fewer bonds to oxygen, or form more bonds to hydrogen. It is almost reversible in some cases to form back oxidation reactions.
A reduction reaction is shown when the reactant side shows a hydrogen within brackets.
Note the similarity between oxidation and reduction in which alcohols can form aldehydes through oxidation and through reduction reverse back into alcohols.
A reduction reaction is shown when the reactant side shows a hydrogen within brackets.
Note the similarity between oxidation and reduction in which alcohols can form aldehydes through oxidation and through reduction reverse back into alcohols.
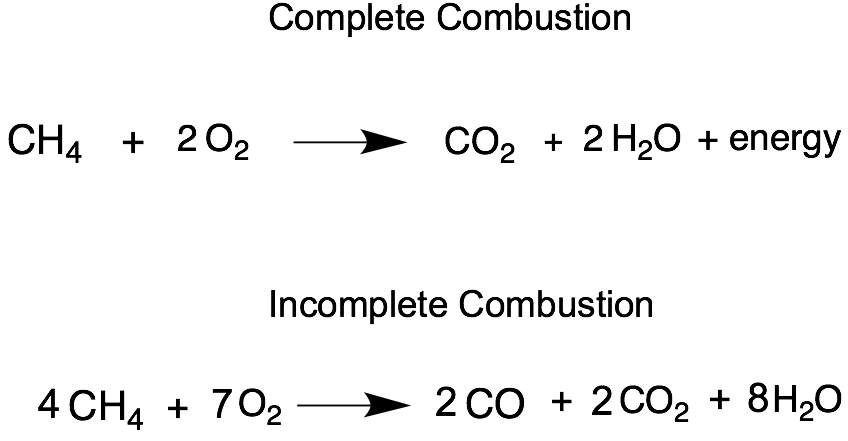
Combustion Reaction
A combustion reaction is not necessarily one that needs to include fire and an explosion. It is just one that includes the reaction between a reactant an oxygen forming carbon dioxide, water and releasing energy.
During an incomplete combustion there is soot, carbon monoxide, carbon dioxide, water and energy that is released. However, an incomplete combustion is much more inefficient than a complete combustion as not all energy from the hydrocarbon is used up.
During an incomplete combustion there is soot, carbon monoxide, carbon dioxide, water and energy that is released. However, an incomplete combustion is much more inefficient than a complete combustion as not all energy from the hydrocarbon is used up.
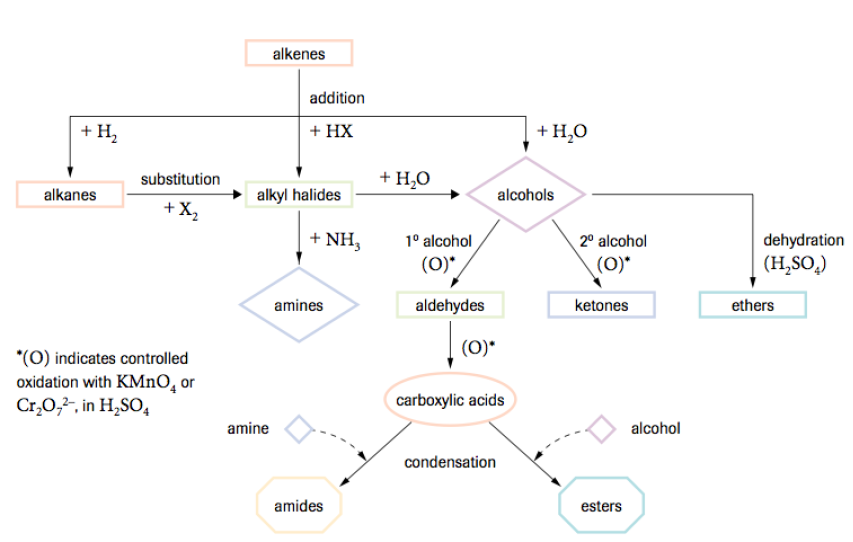
Chapter Summary
These are the different reactions it takes to get from one form of a molecule to another.