Ketones, amines and amides
Ketones
Ketones are distinguished from other chemical compounds from their single C=O bond. It can be easily identified in a hydrocarbon chain as the only double bonded oxygen to a carbon. They are often found in sugars, and compounds that are used in pharmacy. They have a special property that can dissolve many compounds that are insoluble in water, they can remove nail polish, ink stains and many other substances on glass, metal or porcelain objects. The difference between the aldehyde an a ketone is that the aldehyde has the C=O bond on the terminal carbons while the ketone has the C=O anywhere else other than the terminal ends of the hydrocarbon chain. This makes the smallest number of carbons in the ketone three because there must be at least two terminal carbons and one central one.
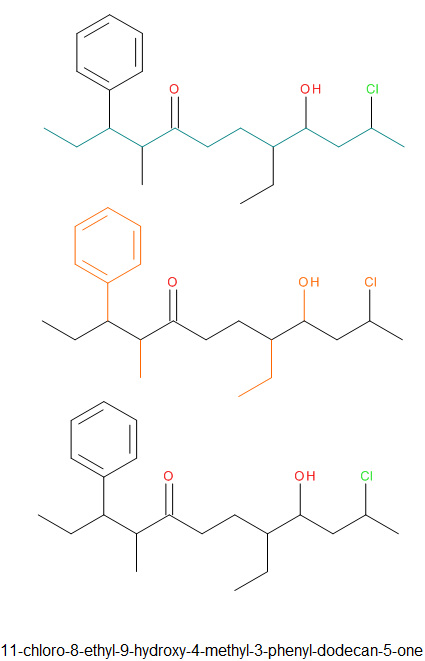
Naming Ketones
|
Properties of Ketones
|
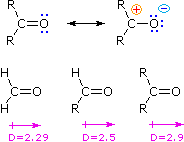
Similar to their aldehyde counterpart, the ketone group also has a carbonyl group which makes it polar. However, this polarity does not allow it to hydrogen bond with other ketones, because there is no hydrogen for the oxygen to have an interaction with. This does not mean that ketones cannot hydrogen bond with other molecules. In fact, ketones are soluble in water because of the hydrogen bonding that they can create. The oxygen atom on the ketone will interact with the hydrogen atom on the water molecule and so securing a bond.
This bond is unique because as the molecule becomes larger and larger the other hydrocarbons get in the way of the hydrogen bonding and lowers solubility in water. |
Amines
|
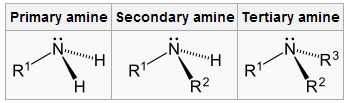
Amines are unique in which it can be either a primary, secondary or tertiary amine just like alcohols. This means that a primary amine is connected to two hydrogens and an R group. The secondary amine is connected to one hydrogen and an R, and R' group. The tertiary amine is connected to an R, R' and R" group. In addition to this, amines also have practical uses, such as rust and corrosion proof coating in boilers.
|
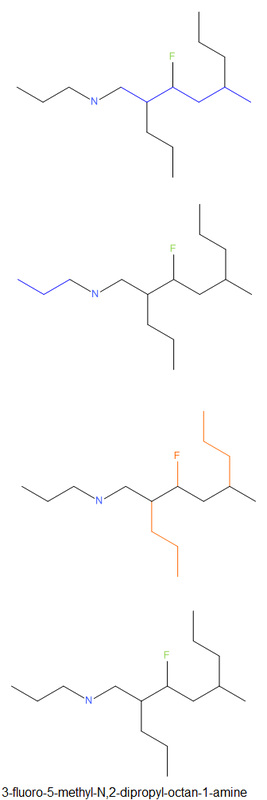
Naming Amines
|
Properties of Amines
|
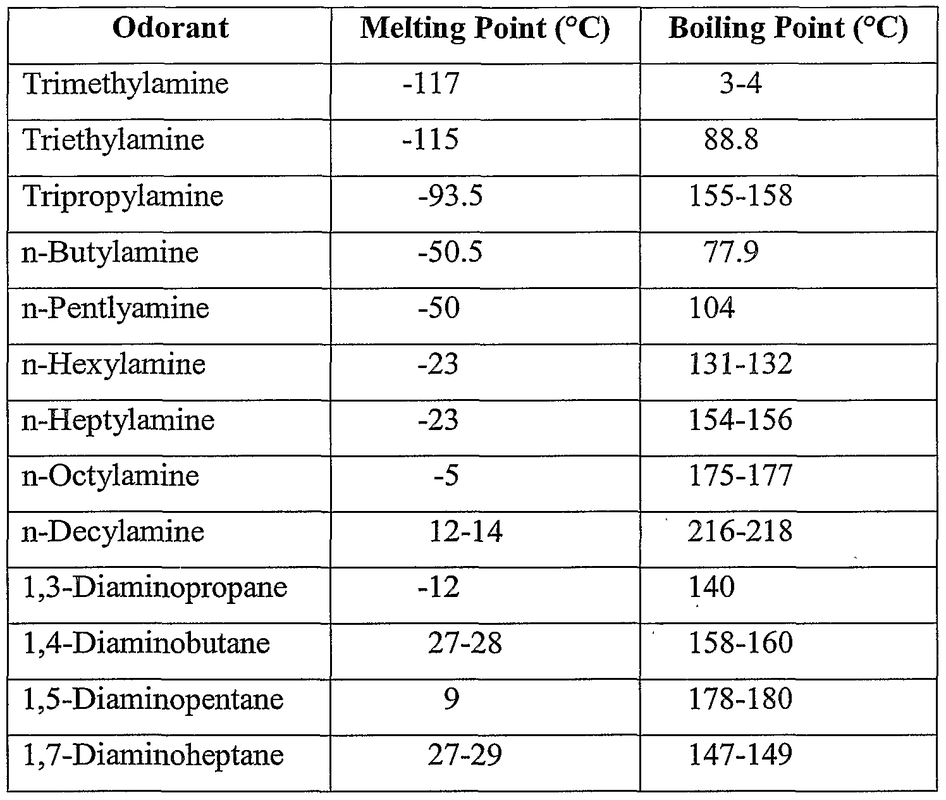
Since the N-H bond is a polar bond there can be hydrogen bonding within and with other molecules. However, this bonding can only happen if there is a primary of secondary amine because there must be a hydrogen connected to the nitrogen and the tertiary amine does not. With the presence of the hydrogen bond, amines can dissolve polar molecules readily and is highly soluble in water as long as there is a hydrogen bonding. As you can guess, the more hydrogen bonds there are the more readily they can dissolve in water and so primary amines are more soluble in water that secondary amines and tertiary amines are not soluble at all.
This polarity also gives it a high boiling point. From the right, the trimethylamine has three carbons bonded to it and so it has a low boiling point. When it loses a carbon chain it's boiling point jumps 80 degrees and then another 80 degrees when it becomes a primary amine. Amines are most notably found in fish in which they give off that fish odor which is the result of amines. |
Amides
|
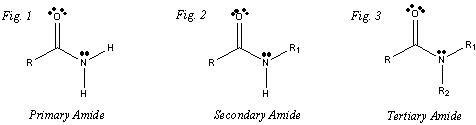
Amides are denoted by their functional group that contains -CON-. It is a carbon double bonded to an oxygen and single bonded to a nitrogen. It is very similar to the ester in which the oxygen is replaced with a nitrogen on the amide. This amide is also separated into three categories: primary, secondary and tertiary. Primary is where the group is connected to one carbon chain, secondary is connected to two carbon chains and the tertiary is connected to three carbon chains. Amides are most notable used as an organic solvent in industry and are used in pain killers.
|
Naming Amides
|
Properties of Amides
|
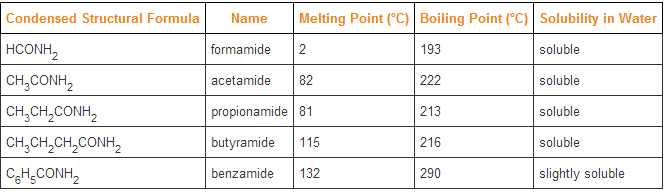
Amides are neutral as opposed to their amine counterpart which is basic. As with their boiling points, amides have an extremely high boiling points when they have longer carbon chains because they can bond with each other nicely with the amounts of hydrogens and nitrogens they have. From the right, it can be seen that the melting points and boiling points continue to rise together as the chains get longer from formamide to benzamide.
However, as the chain gets longer, their solubility in water also drops because of the difference in shape between the two molecules and so not as much hydrogen bonding can occur leading to a smaller solubility. |
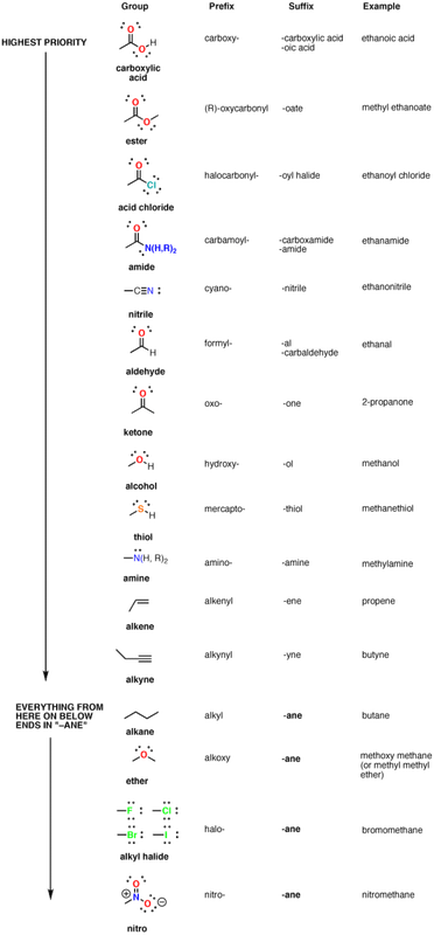
Other Derivatives
Sometimes there may be other functional groups that are in the carbon chains. There must be an order of precedence and below you will see a chart of it.
Chapter Summary
At the very end of it all the polarity is what determines the melting points and boiling points of functional groups. The summary is as follows:
Amide>Acid>Alcohol>Ketone~Aldehyde>Amine>Ester>Ether>Alkane
Amide>Acid>Alcohol>Ketone~Aldehyde>Amine>Ester>Ether>Alkane