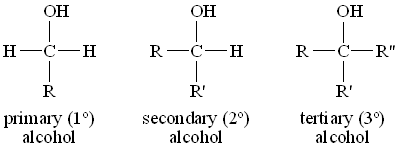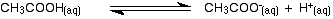Alcohols, Haloalkanes, and Carboxylic Acids
Alcohols
|
Alcohol is a hydrocarbon derivative that contains a hydroxyl functional group. It is an organic compound that contains the -OH functional group. Depending on the position of the hydroxyl group, an alcohol can be primary, secondary, or tertiary. Some common alcohols are methanol, ethanol, propan-2-ol, and ethane-1,2-diol.
Alcohols are very useful as an antiseptic and are often used as an additive to fuel, component of antifreeze and solvent for chemical liquids. |
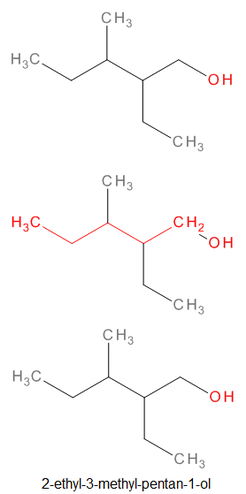
Naming Alcohols
|
Properties of Alcohols
|
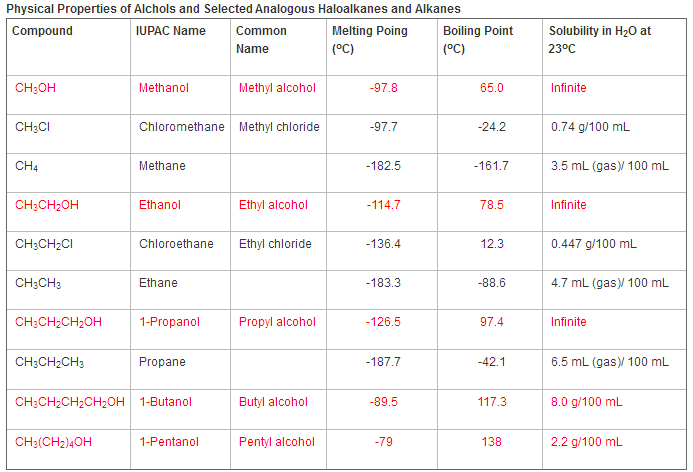
The polarity of the alcohol molecule comes from the hydrogen bonding between the oxygen atom and the hydrogen atom that it is connected to. As a result, the smaller chains of alcohols are miscible with water and can be mixed in any proportions with all amounts being soluble. When there are longer chains of alcohols, the non-polar sides of the alcohol override the smaller polar parts of the molecule and thus make the whole molecule non-polar instead. Since the smaller molecules have a stronger polarity and dissolve readily in water because of the hydrogen bonding. The boiling and melting points are much higher than those that have longer carbon chains.
Facts:
|
Haloalkanes/ Alkyl Halides
|
Alkanes with at least one halogen (F, Cl, Br, I) are named using prefixes: fluoro-, chloro-, bromo-, iodo-. Thesese Haloalkenes are not part of living systems but are synthetic and artificial. CFC or chlorofluorocarbons were used for refrigerants and aerosols sprays but are now much reduced in production and in use because of the damage it causes to the ozone layer. Recently, these CFC's have damaged much of the ozone layer above the Arctic Circle and have allowed harmful ultraviolet rays to enter our atmosphere and so causes more of a problem for global warming.
|
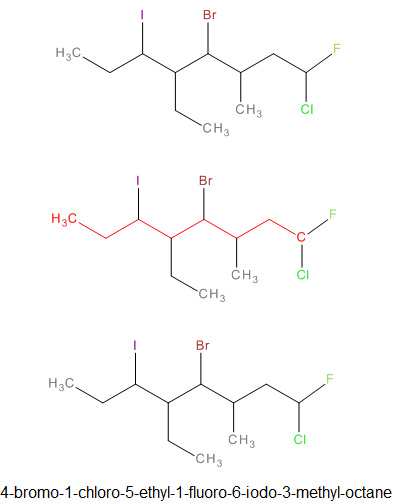
Naming Haloalkanes
|
Properties of Haloalkanes
|
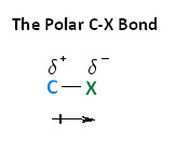
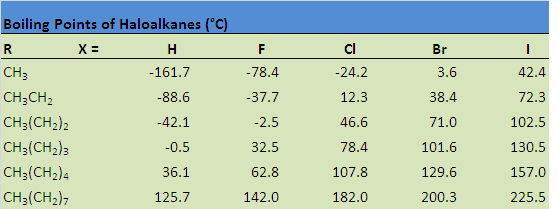
As similar to the alkanes, the smaller groups of carbon such as methyl are soluble in water. The larger they are the less soluble they become. All other groups with the exception of methyl groups are insoluble in water. With haloalkanes, the larger the molecule the higher the boiling point is for that specific molecule. The reason for this is because of the intermolecular forces between the molecules. Firstly on a haloalkane, one of the hydrogen is replaced with a halogen meaning there is an electronegativity difference for the more electronegative atoms such as fluorine and chlorine. As a result, there will be a delta positive and delta positive on the atom. Consider the diagram below:
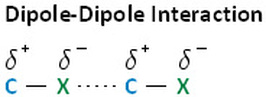
As a result from the polar bond that is created, there is a dipole-dipole interaction between the two molecules if they are stacked on top of each other in a certain orientation. This causes a strong intermolecular bond between the two molecules. In addition to the dipole-dipole interaction, the larger the molecule is the larger the intermolecular force and London Dispersion force because of the high number of electrons and the increased probability of them inducing a charge.
|
Carboxylic Acids
The carboxylic acids is derived from a carboxyl group. Basically it is a carboxyl (double bonded O) group with a hydroxyl (OH) goup attached to it. They are often found in sour foods such as vinegar and oranges. In addition, carboxylic acids are weak acids, unlike their counterpart of HCl where it is a strong acid.
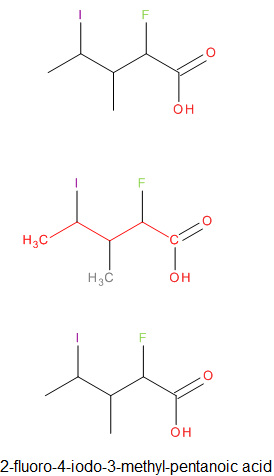
Naming Carboxylic Acids
|
Properties of Carboxylic Acids
|
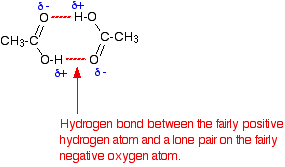
The physical properties of carboxylic acids are very unique in the sense that they have higher boiling points than Alcohols because in pure carboxylic acid, hydrogen bonding can occur between two molecules and thus produce a dimer. This seemingly small increase in size largely increases the intermolecular forces by increasing the London Dispersion forces between the dimer and the surrounding molecules, resulting in a higher boiling point. As you can see on the right, the picture shows you the strong hydrogen bonding that results from the Oxygen and Hydrogen atoms of the molecule. As a result of the shape of the molecule, the hole in the middle will increase the London dispersion forces.
In addition to their strong intermolecular bonds, carboxylic acids are also weak acids and the reactions are reversible. |